What is Cdk2 and why is it an attractive anti-cancer target?
Cyclin-dependent kinase 2 (Cdk2) plays an important role in the cell cycle, regulating the transition from the G1 phase where the cell grows and certain intracellular components (such as the centrosomes) undergo replication, to the S phase, where DNA is replicated. Abnormalities in cell cycle regulation (and CDK activity in particular) is one of the hallmarks of cancer, resulting in the uncontrolled proliferation of cells. Thus, small, selective molecule inhibitors of CDKs are an attractive therapeutic target in the fight against cancer (1-3).
Using Nanolive imaging to investigate how cells respond to Cdk2 inhibition
One of the major advantages of label-free imaging is that images can be acquired at high frequency, for long periods of time. Here, we use exposed glioblastoma (cell line: LN71) and 3T3-derived pre-adipocyte cells to a Cdk2 Inhibitor III and imaged their response with high temporal resolution (one image acquired every 2 secs for 20 h).
This inhibitor targets Cdk2/A, Cdk2/E, Cdk1/B and Cdk4/D1 by acting as a competitive inhibitor for ATP (Fig. 1). The resulting images were stitched and analysed using tools developed in house. In the video below, the graph on the right shows how the average dry mass of a cell changes throughout the experiment; pre-adipocyte cells are shown in green, and glioblastoma cells are shown in orange.
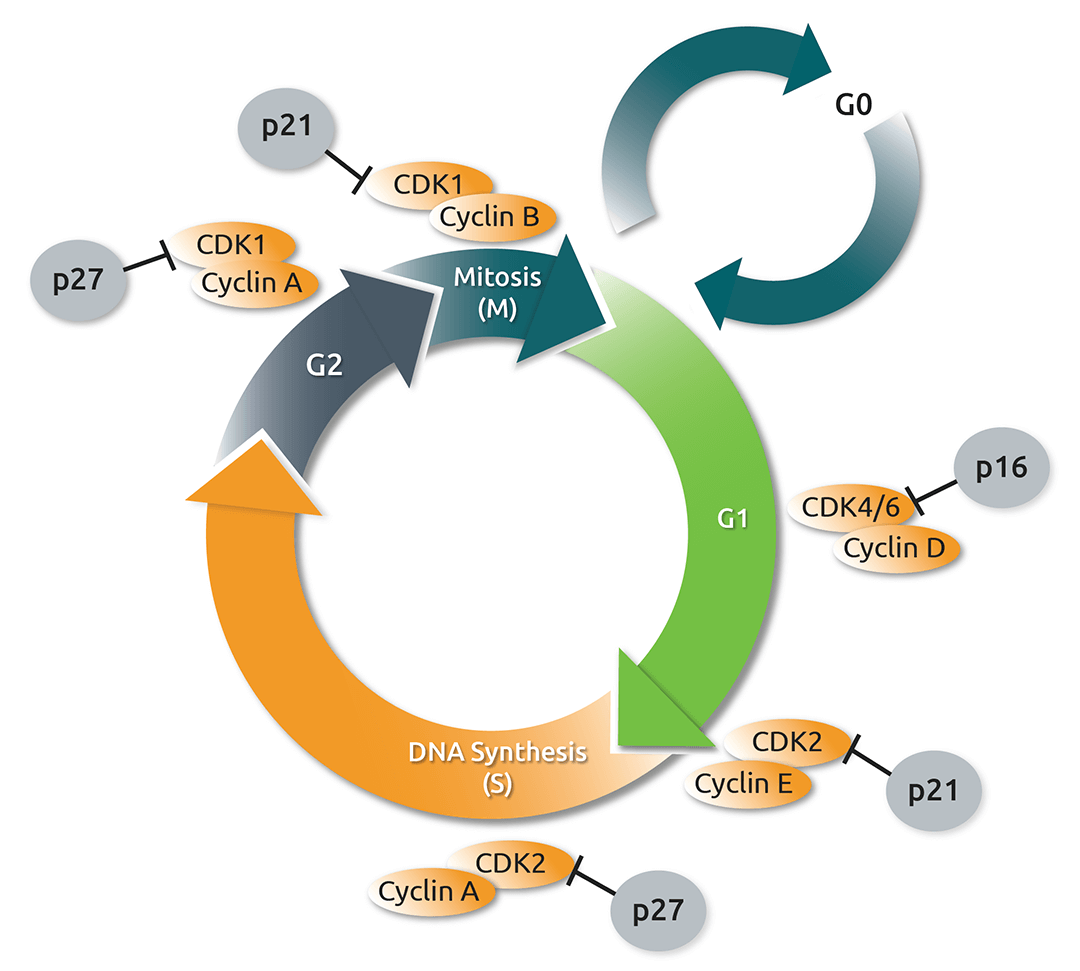
Fig. 1.
A schematic view of the role that cyclin-dependent kinases (CDKs), their regulatory partner proteins (the cyclins), and CDK inhibitors, play in the cell cycle. Figure adapted from (4).
How do pre-adipocytes respond to Cdk2-inhibition?
Initially, Cdk2 inhibition increases the number of protrusions in pre-adipocyte cell and the cells become more spread across the surface of the dish. This response lasts approximately 11 h. At 11:48, the first instance of cell death occurs. After this point, cells start to detach from the surface, lose dry mass and become rounder. There are clear changes in the nuclear membrane, which appears to thicken. A couple of cells die before the experiment is ended, but many remain alive.
Is the response of glioblastoma cells to Cdk2-inhibition comparable?
In short, no. Glioblastoma cells were heavily impacted from the start. No cell protrusions are visible and apoptotic bodies are present in some cells, very early on. Cells start to die 6 h after the inhibitor was added. Death occurs during mitosis, which makes sense considering its role in the cell cycle. Glioblastoma is a very aggressive type of cancer, which divides rapidly, and this may be one reason why we observe mass cell death, and a sharp decrease in dry mass, around the 7-h mark. Some cells survive until the end of the experiment but not many. Surviving cells have a similar phenotype to the remaining pre-adipocyte cells.
References
(1) Asghar U. et al. Nat. Rev. Drug Discov. 14 (2); 130-146 (2015).
(2) Whittaker SR. et al. Pharmacol. Ther. 1 (173); 83-105 (2017).
(3) Otto T. & Sicinski P. Nat. Rev. Cancer. 17 (2); 93-115 (2017).
(4) Bai J. et al. Cancer Biol. Med. 14 (4); 348-362 (2017).
Read our latest news
Revolutionizing lipid droplet analysis: insights from Nanolive’s Smart Lipid Droplet Assay Application Note
Introducing the Smart Lipid Droplet Assay: A breakthrough in label-free lipid droplet analysis Discover the power of Nanolive's Smart Lipid Droplet Assay (SLDA), the first smart digital assay to provide a push-button solution for analyzing lipid droplet dynamics,...
Food additives and gut health: new research from the University of Sydney
The team of Professor Wojciech Chrzanowski in the Sydney Pharmacy School at the University of Sydney have published their findings on the toxic effect of titanium nanoparticles found in food. The paper “Impact of nano-titanium dioxide extracted from food products on...
2023 scientific publications roundup
2023 has been a record year for clients using the Nanolive system in their scientific publications. The number of peer-reviewed publications has continued to increase, and there has been a real growth in groups publishing pre-prints to give a preview of their work....
Nanolive microscopes

CX-A
Automated live cell imaging: a unique walk-away solution for long-term live cell imaging of single cells and cell populations

3D CELL EXPLORER-fluo
Multimodal Complete Solution: combine high quality non-invasive 4D live cell imaging with fluorescence

3D CELL EXPLORER
Budget-friendly, easy-to-use, compact solution for high quality non-invasive 4D live cell imaging



