We congratulate Carlos Tristan, Ilyas Singeç, and the Stem Cell Translation Laboratory team at the National Institutes of Health, USA, on their newly published protocol for single cell cloning; ‘Efficient and safe single-cell cloning of human pluripotent stem cells using the CEPT cocktail’. Nanolive imaging was used to follow the health of individual stem cells and confirmed that the authors’ methods effectively minimised cellular stress and damage.
Visual Signs of Cellular Stress
This paper builds on the team’s previous work to reduce stem cell stress by creating and testing a newly developed small molecule cocktail named CEPT1. Nanolive imaging was used to follow the health of individual cells after dissociation and plating. Cells were imaged shortly after plating, and within the first minutes, the differences between treatments were already detectable. In the absence of the CEPT cocktail, cell stress presented visually as blebbing (rounded bulging of the cell membrane due to uncontrolled cell contractions), abnormal cellular morphologies, and overall reduction in cell roundness as quantified by live-cell imaging.
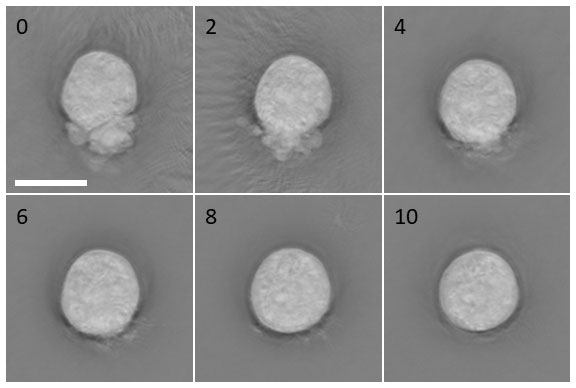
Cell morphology of dissociated hPSCs after treatment with CEPT. Nanolive label-free live cell imaging showing the first 10 min after plating dissociated WA09 cells. Fast-acting CEPT mitigates cell stress and abnormal phenotypes caused by cell contractions and membrane blebbing. Scale bar, 10 µm. Figure kindly provided by C. Tristan.
We were delighted to perform label-free imaging of human pluripotent stem cells for critical experiments using a Nanolive microscope, which enables practical real-time analysis of cell morphology and provides new insights into the dynamic nature of cellular structures.
Automated Imaging Supports Cell Cloning
In their most recent paper2, Tristan et. al standardised the procedure for subcloning single stem cells in order to improve efficiency and survival rates of sensitive cells. After plating cells using a microfluidics system, each 1µl droplet in a 96-well plate must be closely observed; first to check that each droplet contains only one cell; and then to make sure that each cell is healthy and dividing. The authors recommend imaging every 6 hours for up to 14 days which is a significant manual load when using conventional microscopy. With a different Nanolive platform, the CX-A, you don’t need to manually check every well; the automated system will scan the plate, and EVE Analytics will count the number of cells in each well for you.
References
[1] Chen, Y. et al. A versatile polypharmacology platform promotes cytoprotection and viability of human pluripotent and differentiated cells. Nat. Methods 18, 528–541 (2021).
[2] Tristan, C.A. et al. Efficient and safe single-cell cloning of human pluripotent stem cells using the CEPT cocktail. Nat Protoc, (2022)
Read our latest news
Cytotoxic Drug Development Application Note
Discover how Nanolive’s LIVE Cytotoxicity Assay transforms cytotoxic drug development through high-resolution, label-free quantification of cell health and death. Our application note explores how this advanced technology enables real-time monitoring of cell death...
Investigative Toxicology Application Note
Our groundbreaking approach offers a label-free, high-content imaging solution that transforms the way cellular health, death, and phenotypic responses are monitored and quantified. Unlike traditional cytotoxicity assays, Nanolive’s technology bypasses the limitations...
Phenotypic Cell Health and Stress Application Note
Discover the advanced capabilities of Nanolive’s LIVE Cytotoxicity Assay in an application note. This document presents a detailed exploration of how our innovative, label-free technology enables researchers to monitor phenotypic changes and detect cell stress...



