Vesicle Transport
Vesicle transport is an active and thus energy consuming process that the cell uses in order to capture or release macromolecules into or out of the cell.
While exocytosis helps release content like proteins, waste products or toxins to the outside of the cell via vesicles that are formed in the Golgi apparatus and then fused with the plasma membrane, endocytosis aims to capture substances by enclosing them in vesicles resulting from cell membrane folding.
Endocytosis
Depending on the content to engulf, endocytosis can be divided in phagocytosis, receptor-mediated endocytosis, and pinocytosis.
As we showed in our Macrophages – the big eater’s post, phagocytosis is a crucial process in macrophages, as it allows them to get rid of microbes, thus playing a role in cell immunity. This cell eating of solid material results in the formation of phagosomes, which are vesicles coming from the evagination of the cell membrane that will fuse with lysosomes carrying the enzymes required to break the engulfed substances.
When instead of solid particles it is liquid material encapsulated and internalised by the cell we can differentiate between receptor-mediated endocytosis and pinocytosis… but what is the difference between them? While receptor-mediated endocytosis has specificity in the captured substances, pinocytosis is a non-specific cell drinking1.
Pinocytosis
Hence, pinocytosis is the process where fluid matter coming from the outside of the cell is obtained via invaginations of the cell membrane. Its biological meaning is mainly to absorb extracellular fluids (ECF) containing solutes like sugars or proteins, once triggered by the presence of certain substances outside the cell (amino acids or certain ions) but it is also involved in cell immunity.
Two types of pinocytosis can be characterized. Micropinocytosis is observed in cells such as the microvilli of the digestive tract and involves the intake of small vesicles. Vesicles 5 to 50 times bigger than those formed during pinocytosis are seen in macropinocytosis, usually as a result of immune system response in areas where pathogens and antigens are found. A greater deformation of the membrane is needed in order to lead to the formation of big vesicles. As observed in the video, the membrane extends forming arm-like claws that reconnect resulting in a macropinosome (Figure 1). Once at the cytoplasm, the macropinosome fuses with a lysosome2,3.
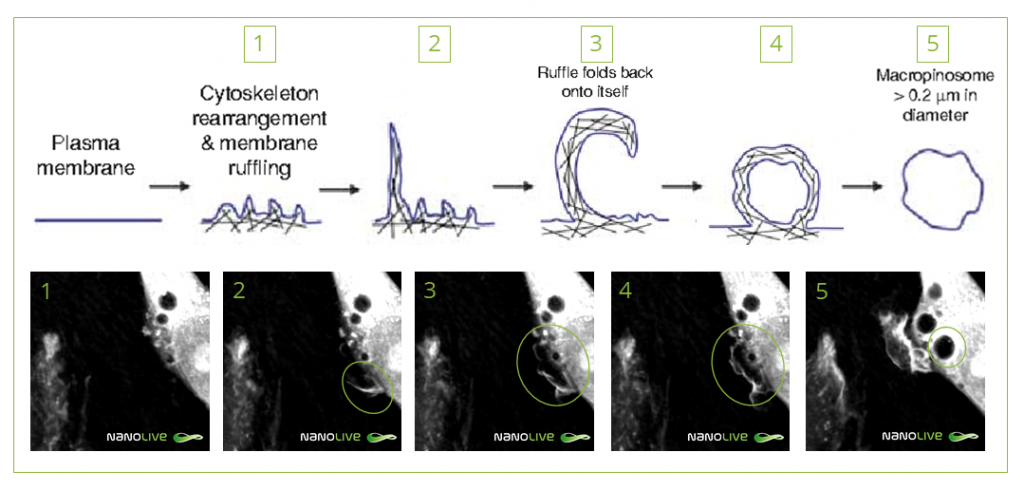
Figure 1. Steps of macropinocytosis. Equivalence between schema and 3D Cell Explorer microscope caption. Schema from: Lim, J.P., & Gleeson, P.A. (2011). Macropinocytosis: an endocytic pathway for internalising large gulps. Immunology and cell biology, 89 8, 836-43.
References
- Ellinger, I. & Pietschmann, P. Endocytosis in health and disease—a thematic issue dedicated to Renate Fuchs. Wiener Medizinische Wochenschrift 166, 193–195 (2016).
- Kruth, H. S. et al. Macropinocytosis Is the Endocytic Pathway That Mediates Macrophage Foam Cell Formation with Native Low Density Lipoprotein. J. Biol. Chem. 280, 2352–2360 (2005).
- Bhat
Read our latest news
Cytotoxic Drug Development Application Note
Discover how Nanolive’s LIVE Cytotoxicity Assay transforms cytotoxic drug development through high-resolution, label-free quantification of cell health and death. Our application note explores how this advanced technology enables real-time monitoring of cell death...
Investigative Toxicology Application Note
Our groundbreaking approach offers a label-free, high-content imaging solution that transforms the way cellular health, death, and phenotypic responses are monitored and quantified. Unlike traditional cytotoxicity assays, Nanolive’s technology bypasses the limitations...
Phenotypic Cell Health and Stress Application Note
Discover the advanced capabilities of Nanolive’s LIVE Cytotoxicity Assay in an application note. This document presents a detailed exploration of how our innovative, label-free technology enables researchers to monitor phenotypic changes and detect cell stress...
Nanolive microscopes

3D CELL EXPLORER
Budget-friendly, easy-to-use, compact solution for high quality non-invasive 4D live cell imaging

3D CELL EXPLORER-fluo
Multimodal Complete Solution: combine high quality non-invasive 4D live cell imaging with fluorescence

CX-A
Automated live cell imaging: a unique walk-away solution for long-term live cell imaging of single cells and cell populations



