Dr. Patrick Sandoz has been collaborating with Nanolive since 2013, first as a PhD student and then as a post-doc in Prof. van der Goot’s lab at EPFL (Lausanne, Switzerland). He played a very important role in testing Nanolive’s first microscope, the 3D Cell Explorer. He kindly agreed to meet with us to discuss his journey with Nanolive, his ground-breaking paper published in the prestigious journal PLOS Biology (see here), and what he thinks Nanolive imaging could be used for in future research.
One of the major advantages of Nanolive imaging is that we can observe contact points between cells, migration patterns and different morphologies, all in an unperturbed system.
“It all began when Nanolive approached my PhD supervisor Prof. van der Goot, and asked whether we could test their first microscope, the 3D Cell Explorer. We imaged some cells and we were pretty sure we could identify lipid droplets and mitochondria from the refractive index (RI) images but to be sure we needed to prove that”. This is exactly what Patrick did two years later, with Masters student Christopher Tremblay, once the 3D Cell Explorer-Fluo had been developed. They designed a proof-of-concept study to correlate RI images with fluorescent images using genetically modified cell lines that express fluorescent proteins in different cellular structures. „We did many tests with Dr. Mathieu Frechin (Head of Quantitative Biology at Nanolive). We tracked lipid droplets in 3D and perturbed the system to see how it behaves. We investigated the changes in dry mass that occur before, during, and after mitosis and we quantified the nuclear rotation that occurs in cells prior to division“. This spinning phenomena in the nucleus is what we can observe in this video (S5 Movie in Sandoz et al., 2019).

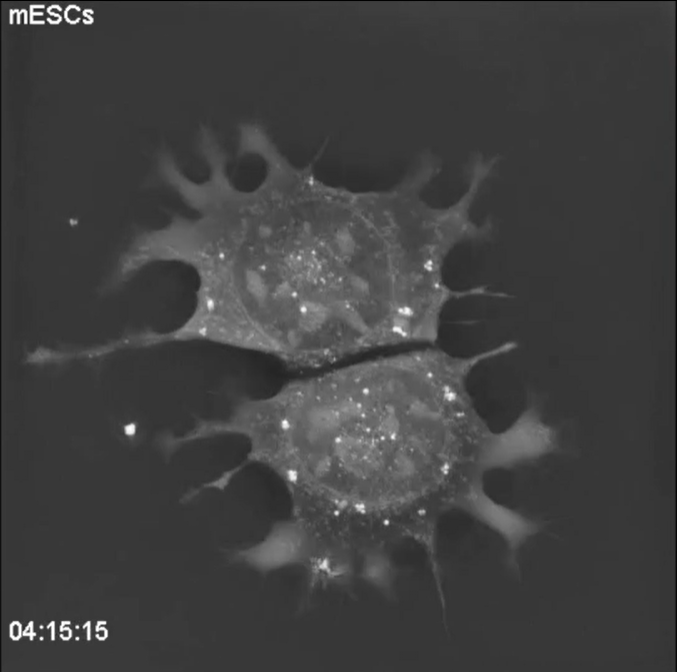
“The big plus of Nanolive imaging is being able to see everything that is going on in the cell, at the same time, without generating phototoxicity.” This lack of phototoxicity means even sensitive cells such as the mouse embryonic stem cells (mESCs) in this
video (S1 Movie in Sandoz et al., 2019), can be observed for long periods of time, at high spatial and temporal resolution. In this footage, which was acquired over a period of 10 h; at a frequency of four images per minute, we observe fine mitochondrial fusion and fission events as well as two full mitosis cycles.
Patrick currently works as a post-doc at the KTH Royal Institute of Technology (Stockholm, Sweden), where he works on primary cytotoxic T lymphocytes, which are notoriously sensitive and challenging to image. Last year he tried to image these cells using the 3D Cell Explorer, with great success. “The cells were very happy; we could observe contact points between cells, migration patterns and different morphologies, all in an unperturbed system”. I ask whether Patrick could envisage an experiment where these types of observations could be useful. “I think there are thousands of projects that this could be applicable to, depending of course, on the focus of the lab in question. It could be very interesting to investigate for example, the polarization of the cell, or the role that refractive fibres play in the remodeling of the cytoskeleton”. This last point has been raised by a lot of our clients recently and so, I decide to ask why. “If you want to study actin or tubulin you always need to have a fluorescent protein expressed but this blocks the polymerization of the tube, resulting in the cell becoming stuck in an unnatural conformation. Using the refractive index keeps the cells in a natural behaviour without perturbing the system, so if you want to add a drug/inhibitor to block a certain pathway you visualize the real response of the cell, rather than the artifact imposed by the fluorescent protein”. Nanolive would like to thank Patrick for taking the time to meet with us and share his very interesting experiences with our microscopes.
Read our latest news
Revolutionizing lipid droplet analysis: insights from Nanolive’s Smart Lipid Droplet Assay Application Note
Introducing the Smart Lipid Droplet Assay: A breakthrough in label-free lipid droplet analysis Discover the power of Nanolive's Smart Lipid Droplet Assay (SLDA), the first smart digital assay to provide a push-button solution for analyzing lipid droplet dynamics,...
Food additives and gut health: new research from the University of Sydney
The team of Professor Wojciech Chrzanowski in the Sydney Pharmacy School at the University of Sydney have published their findings on the toxic effect of titanium nanoparticles found in food. The paper “Impact of nano-titanium dioxide extracted from food products on...
2023 scientific publications roundup
2023 has been a record year for clients using the Nanolive system in their scientific publications. The number of peer-reviewed publications has continued to increase, and there has been a real growth in groups publishing pre-prints to give a preview of their work....
Nanolive microscopes

3D CELL EXPLORER
Budget-friendly, easy-to-use, compact solution for high quality non-invasive 4D live cell imaging

3D CELL EXPLORER-fluo
Multimodal Complete Solution: combine high quality non-invasive 4D live cell imaging with fluorescence

CX-A
Automated live cell imaging: a unique walk-away solution for long-term live cell imaging of single cells and cell populations



